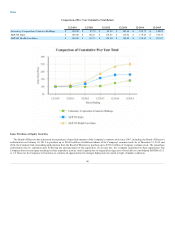
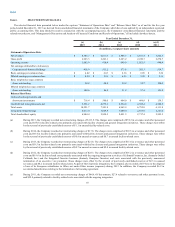
LabCorp 2015 Annual Report Download - page 42
Download and view the complete annual report
Please find page 42 of the 2015 LabCorp annual report below. You can navigate through the pages in the report by either clicking on the pages listed below, or by using the keyword search tool below to find specific information within the annual report.
Index
Acts of fourteen states and the District of Columbia. The lawsuit seeks actual and treble damages and civil penalties for each alleged false claim, as well as
recovery of costs, attorney's fees, and legal expenses. Neither the U.S. government nor any state government has intervened in the lawsuit. The Company's
Motion to Dismiss was granted in October 2014 and Plaintiff was granted the right to replead. On January 11, 2016, Plaintiff filed a motion requesting leave
to file an amended complaint under seal and to vacate the briefing schedule for the Company's Motion to Dismiss, while the government reviews the
amended complaint. The Court granted the motion and vacated the briefing dates. Plaintiff then filed an amended complaint under seal. The Company will
vigorously defend the lawsuit.
In addition, the Company has received various other subpoenas since 2007 related to Medicaid billing. In October 2009, the Company received a
subpoena from the State of Michigan Department of Attorney General seeking documents related to its billing to Michigan Medicaid. In June 2010, the
Company received a subpoena from the State of Florida Office of the Attorney General requesting documents related to its billing to Florida Medicaid. In
October 2013, the Company received a civil investigative demand from the State of Texas Office of the Attorney General requesting documents related to its
billing to Texas Medicaid. The Company is cooperating with these requests.
On November 4, 2013, the State of Florida through the Office of the Attorney General filed an Intervention Complaint in a False Claims Act lawsuit, State
of Florida ex rel. Hunter Laboratories, LLC and Chris Riedel v. Quest Diagnostics Incorporated, et al. in the Circuit Court for the Second Judicial Circuit for
Leon County. The Intervention Complaint, originally filed by a competitor laboratory, alleges that the Company overcharged Florida’s Medicaid program.
The lawsuit seeks actual and treble damages and civil penalties for each alleged false claim, as well as recovery of costs, attorney’s fees, and legal expenses.
The Company's Motion to Dismiss was denied in February 2015. The Company will vigorously defend the lawsuit.
On May 2, 2013, the Company was served with a False Claims Act lawsuit, State of Georgia ex rel. Hunter Laboratories, LLC and Chris Riedel v. Quest
Diagnostics Incorporated, et al., filed in the State Court of Fulton County, Georgia. The lawsuit, filed by a competitor laboratory, alleges that the Company
overcharged Georgia's Medicaid program. The State of Georgia filed a Notice of Declination on August 13, 2012, before the Company was served with the
Complaint. The case was removed to the United States District Court for the Northern District of Georgia. The lawsuit seeks actual and treble damages and
civil penalties for each alleged false claim, as well as recovery of costs, attorney's fees, and legal expenses. On March 14, 2014, the Company's Motion to
Dismiss was granted. The Plaintiffs repled their complaint and the Company filed a Motion to Dismiss the First Amended Complaint. In May 2015, the Court
dismissed the Plaintiffs' anti-kickback claim and remanded the remaining state law claims to the State Court of Fulton County. In July 2015, the Company
filed a Motion to Dismiss these remaining claims. The Plaintiffs filed an opposition to the Company's Motion to Dismiss in August 2015. Also, the State of
Georgia filed a brief as amicus curiae. The Company will vigorously defend the lawsuit.
On February 27, 2012, the Company was served with a False Claims Act lawsuit, United States ex rel. Margaret Brown v. Laboratory Corporation of
America Holdings and Tri-State Clinical Laboratory Services, LLC, filed in the United States District Court for the Southern District of Ohio, Western
Division. The Company owned 50% of Tri-State Clinical Laboratory Services, LLC, which was dissolved in June of 2011 pursuant to a voluntary petition
filed under Chapter 7 of Title 11 of the United States Code. The lawsuit alleges that the defendants submitted false claims for payment for laboratory testing
services performed as a result of financial relationships that violated the Stark and the Anti-Kickback Statute. The lawsuit seeks actual and treble damages
and civil penalties for each alleged false claim, as well as recovery of costs, attorney's fees, and legal expenses. The U.S. government has not intervened in the
lawsuit. The parties have reached a settlement in principle, but the Company will vigorously defend the lawsuit if the settlement is not finalized.
On June 7, 2012, the Company was served with a putative class action lawsuit, Yvonne Jansky v. Laboratory Corporation of America, et al. , filed in the
Superior Court of the State of California, County of San Francisco. The lawsuit alleges that the defendants committed unlawful and unfair business practices,
and violated various other state laws by changing screening codes to diagnostic codes on laboratory test orders, thereby resulting in customers being
responsible for co-payments and other debts. The lawsuit seeks injunctive relief, actual and punitive damages, as well as recovery of attorney's fees, and legal
expenses. In June 2015, Plaintiff's Motion for Class Certification was denied. The Plaintiff has appealed the denial of class certification, and the trial court has
stayed the case pending resolution of the appeal. The Company will vigorously defend the lawsuit.
On August 24, 2012, the Company was served with a putative class action lawsuit, Sandusky Wellness Center, LLC, et al. v. MEDTOX Scientific, Inc., et
al., filed in the United States District Court for the District of Minnesota. The lawsuit alleges that on or about February 21, 2012, the defendants violated the
U.S. Telephone Consumer Protection Act (TCPA) by sending unsolicited facsimiles to Plaintiff and more than 39 other recipients without the recipients' prior
express invitation or permission. The lawsuit seeks the greater of actual damages or the sum of $0.0005 for each violation, subject to trebling under the
TCPA, and injunctive relief. In September of 2014, Plaintiff’s Motion for Class Certification was denied. In January of 2015, the Company’s Motion
42